The Latest News from OACS Ireland
Families urge Donnelly to hold inquiry into use of epilepsy drug during pregnancy.
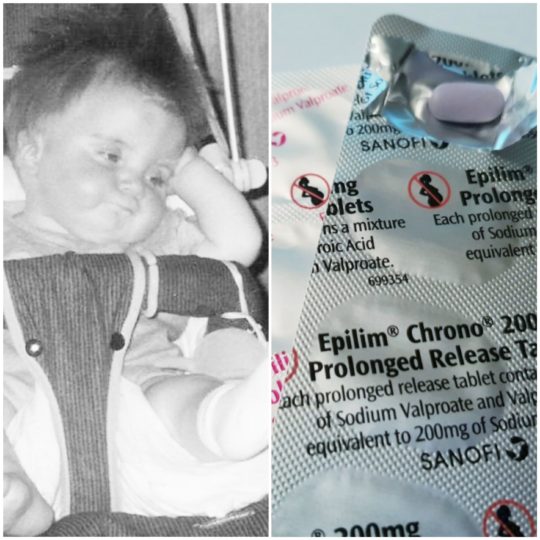
On Saturday the 04/03/2023, Niamh Griffin from the Irish Examiner published yet another outstanding article on the lack of justice for those who have been injured by Epilim (Sodium Valproate) some children and adults who are catastrophically injured. The truth…
More . . .OACS Ireland writes to the Minister for Health Stephen Donnelly on the Epilim (Sodium Valproate) Inquiry.
Re: Sodium Valproate (EPILIM) Inquiry. Dear Minister, We trust this finds you well. We are contacting you to ascertain a precise indication for the establishment of the inquiry into the historical licensing and prescribing of Sodium Valproate in Ireland together…
More . . .Sodium Valproate “Epilim” Campaign Update
Last updated:05/12/2022 We and our colleagues in Epilepsy Ireland are pleased to inform our members and supporters that there have been two positive developments recently regarding our ongoing joint campaign surrounding Sodium Valproate (Epilim). Which are the Nurses and the…
More . . .OACS Ireland Annual Report 2021
Chairs Opening Remarks 2021 was another important year as we sought to progress the Inquiry into the historical licensing and use of sodium valproate (Epilim) in Ireland which was promised by the Minister for Health in November 2020. At the…
More . . .Supporting #MedSafetyWeek 2022
Global campaign to promote reporting side effects from medicines. Takes place from Monday 7 November to Sunday 13 November 2022. OACS Ireland will be supporting the HPRA with their part in the seventh annual #MedSafetyWeek, a global week-long social media campaign to raise…
More . . .Crucial Update on Topiramate (Topamax) for women with epilepsy and other conditions of childbearing potential.
Updated the 24/10/2022 During the summer, a new piece of research suggested that the potential neurodevelopmental impacts associated with the anti-seizure medication (ASM) Topiramate (Topamax) if prescribed during pregnancy may be more significant than previously thought. The paper led to…
More . . .OACS Ireland and Epilepsy Ireland meet with the Minister for Health
A spokesman for OACS Ireland and Epilepsy Ireland, supporting the families, said the meeting took place on Tuesday. “The meeting was a frank exchange of views on the latest proposed terms of reference for the inquiry, between the patient groups…
More . . .A Irish Independent Exclusive. The stories of the families affected by Sodium Valproate (Epilim) in Ireland…
The Irish Independent have published a documentary featuring women who were prescribed Sodium Valproate (Epilim) during pregnancy. The documentary discusses how this has affected their life and the lives of their children. The feature is a powerful piece which highlights…
More . . .OACS Ireland & Epilepsy Ireland to meet with Minister for Health regarding Epilim (Sodium Valproate) Inquiry
Last Updated: Wed, 10/08/2022 OACS Ireland & Epilepsy Ireland will be meeting with the Minister for Health regarding the proposed Terms of Reference into the historical licensing and prescribing of Epilim (Sodium Valproate). This drug can cause a range of disabilities…
More . . .New study published on neurodevelopmental outcomes on prenatal exposure to AEDs
A new study has found that the potential neurodevelopmental impacts associated with the anti-epileptic drug (AED) Topiramate (Topamax) if prescribed during pregnancy may be more significant than previously thought. The study also found increased risks associated with other AEDs when…
More . . .Downloads & Resources
Links to Website Resources
- OACS Facebook Group
- OACS YouTube Channel
- OACS UK Charity
- Epilepsy Ireland
- Irish Epilepsy and Pregnancy Register
- Irish Family Planning Association
- Birth Control (HSE Website)
- Valproate (Epilim) Patient Booklet (HPRA)
- EMA – Information & reviews on Valproate
- Valproate Information(HSE Website)
- Meeting highlights from PRAC
Download Resources
- OACS Ireland Leaflet
- National Conference on the Consequences of Sodium Valproate
- Joint Committee on Health Report on Foetal Anti-Convulsant Syndrome
- Valproate (Epilim) is a medicine licensed in Ireland to treat epilepsy and bipolar disorder
- 2019 Neurology Referral Specialist
- HSE Report on prenatal exposure
- 2018 Sodium Valproate who knew what and when
- 2019 Videos from the National Conference on Sodium Valproate
- 2018 FACS Forum welcomes HSE’s plans to contact patients directly regarding Sodium Valproate
- 2019 Study identifies the risk of malformations associated with prenatal exposure to 10 epilepsy drugs
Videos / Media
OACS Ireland: The Story So Far
Public Hearing on Valproate
Epilim (Sodium Valproate): For Patients*
Epilim (Sodium Valproate): For Hospital Doctors*
Epilim (Sodium Valproate): For Neurologists & Psychiatrists*
Epilim (Sodium Valproate): For GPs*
*The Medication Safety Minute Team in conjunction with Dr Colin Doherty, Epileptologist in St. James’s Hospital (SJH), have produced a series of 4 videos to highlight the risks associated with sodium valproate medicines if taken during pregnancy. Each of the videos highlights key points in relation to the management of the issue from a different perspective − that of the patient, the hospital doctor, the specialist (neurologist or psychiatrist), and the GP, respectively. These videos were released during National Epilepsy Week which aims to raise awareness Epilim (Sodium Valproate) in pregnancy.