Today as part of #EpilepsyWeek, we focus on the need for a Stakeholder Group !
DAY 3 #EpilepsyWeek
Today as part of #EpilepsyWeek, we focus on the need for a stakeholder group to be established by the Minister to review and strengthen the risk reduction measures around Sodium Valproate (Epilim). There are currently approximately 1,700 women and girls between 16-44 years of age taking valproate in Ireland today. We estimate that less than 1 in 3 are on the PPP, three years after it was launched.
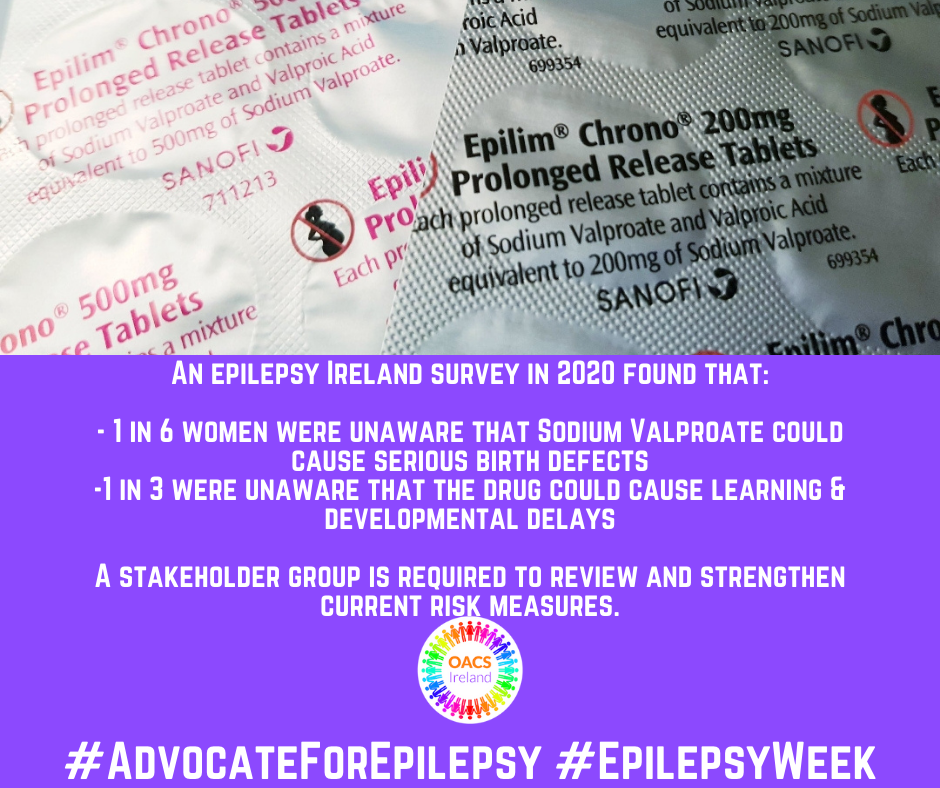
As a result of our campaigning alongside Epilepsy Ireland, a series of measures were introduced in 2014 and again in 2018 – all aimed at ensuring a women on Valproate was fully informed of the risks. These included updated patient information leaflets; new packaging which carried warnings; educational materials for healthcare professionals alongside other measures. The pinnacle of all this was what we call the Pregnancy Prevention Programme. This dictated that women who are prescribed the drug would be seen yearly and would be asked to sign an annual risk acknowledgement form – again all these measure were aimed at ensuring that those on the drug were fully informed of the risks.
However, an Epilepsy Ireland survey last year found that women on Valproate were still dangerously unaware of the risks and indeed, of the measures that are supposed to be applied around the prescribing and distribution of the medication.
Therefore, the current measures that are in place urgently need to be revisited – to ensure that no other family is affected. We have called for a Ministerial Stakeholder Group to be established – which would involve patient organisations and all those who would be involved in the care of women with epilepsy and other conditions. Therefore the immediate establishment of a Valproate Stakeholder Group reporting to the Minister to address these concerns through a co-ordinated and comprehensive approach. Stakeholders should include the HSE, HPRA, Pharmaceutical Society of Ireland, Irish Pharmacy Union, ICGP etc and patient groups.
The Minister has agreed to such a measure however, there has been no details provided from the DOH since our meeting in November. Mean while – some women are going uninformed of the risks which Epilim (Sodium Valproate) poses in pregnancy. We are calling for this group to be set up with urgency to ensure no other family will be impacted by Epilim (Sodium Valproate) in the future.
#AdvocateForEpilepsy this #EpilepsyWeek and contact your local TD or Senator in support of our calls !
https://www.epilepsy.ie/content/sodium-valproate-epilim-campaign-0
